- Blog
- Direct X Windows 7 Manual Download
- Casio Module No.2747 User's Manual
- Mirro 22 Quart Pressure Canner User Manual
- Pelonis Heater Hf 0063 User Manual
- 2001 Tundra Repair Manual Download
- Casio Tide Graph Watch User Manual
- Evinrude Outboard Manual Free Download 85etlr77s
- Download Owners Manual For Cars
- Ninja Air Fryer Af101 User Manual
- Brookstone Pure Ion Pi-02 User Manual
- Prestige Induction Cooktop Pic 16.0 User Manual
- Bajujet Manual Pdf Download
- Ge Networx Nx 8 User Manual
- Download Vizio Sb4051 Manual
- Hp Officejet 6500 Printer User Manual
- 4r100 Rebuild Manual Download
- User Manual For Samsung Galaxy Tab A 10.5
- Gtn 750 Manual Download
- Allen Bradley Soft Starter User Manual
- 1999 Toyata Tacoma Manual Transmission User Manual
- 2011 Ford Explorer Limited User Manual
- International Function Point Users Group Manual
- Ck100 Auto Key Programmer User Manual
- Datacolor Spyder 4 Pro User Manual
- Blue Point Eecr 1a Manual Download
- Canon Eos 80d User Manual English
- Dynacord Powermate 1000 2 User Manual
- Bosch Silence Plus Dishwasher User Manual
- Alsport Trisport Manual Download
- Samsung Ln40b650 Manual Download
- 20000mah Mi Power Bank 2i User Manual
- Sencore Pa81 User Manual Download
- Inspiron N7010 Manual Free Download
- How To Record With Mustek Dv526l User Manual
- Lg Model Lmx25981st 01 User Manual
- Bactec Fx System User's Manual
- Echo Pb 251 Gas Blower User Manual
- Cricut Expression 2 User Manual Pdf
- Bactec Fx Procedure Manual
- Bactec Fx Instrument Manual
- Bd Bactec Fx40 User Manual
- Bd Bactec Fx User Manual

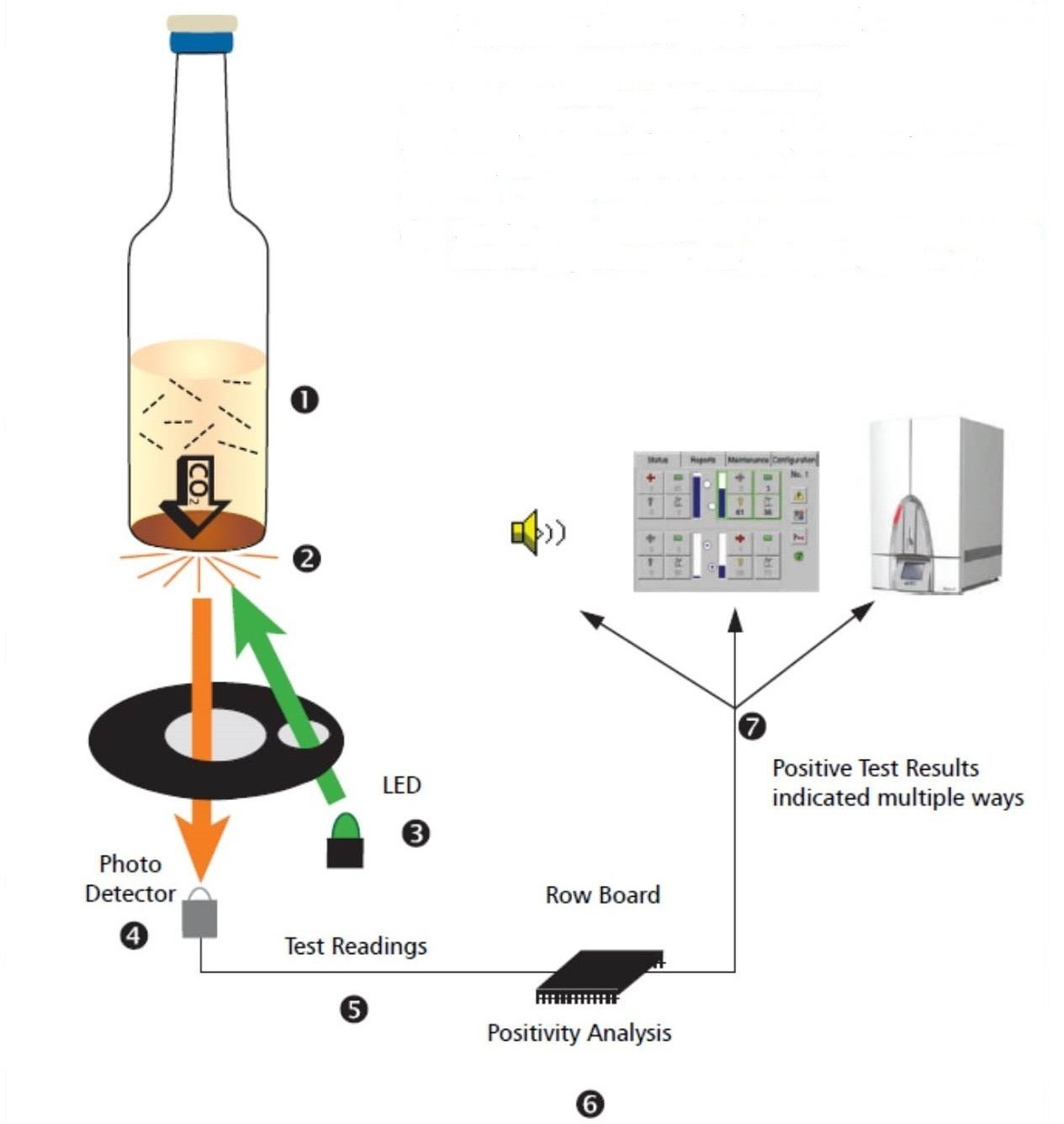
FX2N-32CCL-NP CC-Link SYSTEM LOCAL BLOCK HARDWARE MANUAL JY992D96001A This manual contains text, diagrams and explanations which will guide the reader in the correct installation. Further information can be found in the FX series PLC hardware manuals. BACTEC™ Fluorescent Series User’s Manual 2. BD BACTEC™ 9000 Systems: A World of Difference in Blood Culture Page 3 07/2014 Figure 2 Fluorescent Technology and Sophisticated Algorithms: a Powerful Combination BMS DIAGNOSTICS (M) SDN BHD (485573-V) 19, Jalan 4/62A, Bandar Menjalara, Kepong, 52200 Kuala Lumpur, Malaysia.
PMID: 7494044
The BD BACTEC FX, latest among the bactec blood culture system, is a fully automated microbiology growth and detection system designed to detect microbial growth from blood specimens. It uses fluorescent technology with an exceptional performance, improving workflow efficiencies from specimen collection to actionable results. The BACTEC 9240 blood culture system (Becton Dickinson Diagnostic Instrument Systems, Sparks, Md.) is one of three automated, continuous-monitoring systems that is widely used in clinical laboratories. The BACTEC 9240 was compared with the BACTEC NR 660 for the detection of organisms and bacteremic. BD Diagnostics: Bactec™ FX and BD MAX12 months. 9240 for the same location 441398 FX Software and CD Users Manual, 441510 CD for Use. Currently, both manual and automated blood culture systems are available, with the market being dominated increasingly by instrument-based automated. BACTEC 9240 system (Becton, Dickinson Co. Mar 25, 2019 BACTEC™ FX40 SYSTEM SERVICE TRAINING MODULE B MODULE B 3 Topic 1: System Overview The FX40 instrument is a BACTEC FX derivative instrument design for price sensitive, low volume emerging market labs. The instrument will leverage the design of BACTEC FX measurement system and FX “Vial Activated Workflow”.
This article has been cited by other articles in PMC.
Abstract
The BACTEC 9240 blood culture system (Becton Dickinson Diagnostic Instrument Systems, Sparks, Md.) is one of three automated, continuous-monitoring systems that is widely used in clinical laboratories. The BACTEC 9240 was compared with the BACTEC NR 660 for the detection of organisms and bacteremic episodes; time to detection of positive cultures; number of false-positive and false-negative cultures; and time needed to load, process, and perform quality control functions by using high-volume aerobic media. Blood specimens (5,282) were inoculated in equal volumes (5 to 10 ml per bottle) into BACTEC Plus Aerobic/F (9240 system) and BACTEC Plus NR26 (660 system) bottles. Clinically significant isolates were detected in 6.6% of cultures, representing 348 microorganisms and 216 bacteremic episodes. Two hundred forty-eight microorganisms were detected by both systems, 48 by the 9240 only and 52 by the 660 only (P = not significant). Of the bacteremic episodes, 158 were detected by both systems, 27 by the 9240 only and 31 by the 660 only (P = not significant). Analysis of data by month revealed equivalent recovery rates for both systems, with the exception of a 30-day period at one study site during which the 660 system detected significantly more microorganisms. Following a proprietary hardware design retrofit of the 9240 instrument, detection rates were again equivalent for the remaining three months at this study site. Positive cultures detected by both systems were detected an average of 4.3 h faster by the 9240 system (21 versus 25.3 h). The numbers of false-positive cultures for the 9240 and 660 systems were 40 (1.0%) and 9 ( < 1.0%), respectively.(ABSTRACT TRUNCATED AT 250 WORDS)
Full Text
The Full Text of this article is available as a PDF (207K).
Bactec Fx Procedure Manual
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
Bactec Fx Instrument Manual
- Morello JA, Leitch C, Nitz S, Dyke JW, Andruszewski M, Maier G, Landau W, Beard MA. Detection of bacteremia by Difco ESP blood culture system. J Clin Microbiol. 1994 Mar;32(3):811–818.[PMC free article] [PubMed] [Google Scholar]
- Nolte FS, Williams JM, Jerris RC, Morello JA, Leitch CD, Matushek S, Schwabe LD, Dorigan F, Kocka FE. Multicenter clinical evaluation of a continuous monitoring blood culture system using fluorescent-sensor technology (BACTEC 9240). J Clin Microbiol. 1993 Mar;31(3):552–557.[PMC free article] [PubMed] [Google Scholar]
- Wasilauskas B, Gay R, Zwadyk P, Pfaller M, Koontz F. Multicenter comparison of MicroScan and BACTEC blood culture systems. J Clin Microbiol. 1987 Dec;25(12):2355–2358.[PMC free article] [PubMed] [Google Scholar]
- Wilson ML, Weinstein MP, Reimer LG, Mirrett S, Reller LB. Controlled comparison of the BacT/Alert and BACTEC 660/730 nonradiometric blood culture systems. J Clin Microbiol. 1992 Feb;30(2):323–329.[PMC free article] [PubMed] [Google Scholar]
- Zwadyk P, Jr, Pierson CL, Young C. Comparison of Difco ESP and Organon Teknika BacT/Alert continuous-monitoring blood culture systems. J Clin Microbiol. 1994 May;32(5):1273–1279.[PMC free article] [PubMed] [Google Scholar]
Articles from Journal of Clinical Microbiology are provided here courtesy of American Society for Microbiology (ASM)
-
|
Bd Bactec Fx40 User Manual
Bd Bactec Fx User Manual
| ||||||||||||||||||||||||||||||||||
